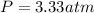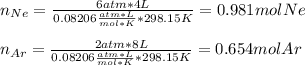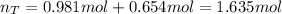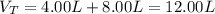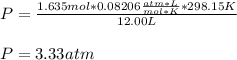Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 00:30
Butadiene undergoes a reaction at a certain temperature in the gas phase as follows: 2c4h6(g) --> c8h12(g) the following data were collected for this reaction: time (min) [c4h6] (m) 0 0.36 15 0.30 30 0.25 48 0.19 75 0. determine the order of the reaction and the rate constant. 1st order and k = 4.3x10 -4 s-1 1st order and k = 2.3x10-4 s-1 2nd order and k = 4.3x10-4 s-1 2nd order and k = 2.3x10-4 s-1 zero and k = 4.3x10-4 s-1
Answers: 3

Chemistry, 22.06.2019 02:00
Which of the following happens during cell division? (a) energy is created (b) waste is eliminated (c) carbon dioxide is released (d) damaged cells are replaced
Answers: 1

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1
You know the right answer?
A 4.00L flask containing Ne at 25 C and 6.00 atm is joined by a valve to an 8.00 L flask Ar at 25 C...
Questions


Advanced Placement (AP), 13.01.2021 21:40





Mathematics, 13.01.2021 21:40

Arts, 13.01.2021 21:40

Mathematics, 13.01.2021 21:40

Physics, 13.01.2021 21:40


Computers and Technology, 13.01.2021 21:40


Social Studies, 13.01.2021 21:40


Mathematics, 13.01.2021 21:40

History, 13.01.2021 21:40

Mathematics, 13.01.2021 21:40


English, 13.01.2021 21:40