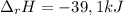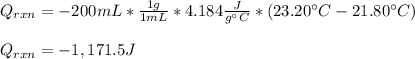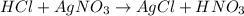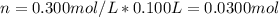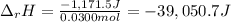100 mL of a 0.300 M solution of AgNO3 reacts with 100 mL of a 0.300 M solution of HCl in a
coffee-cup calorimeter and the temperature rises from 21.80 °C to 23.20 °C. Assuming the density
and specific heat of the resulting solution is 1.00 g/mL and 4.18 J/g. °C, respectfully, what is the
AHºx?
A 39.0 kJ/mol
B +39.0 kJ/mol
C.+1.17 kJ/mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Rocks, as they are compressed, begin forming mountains above the earth's surface when two continental plates converge. the continental crust increases in depth as the mountains grow above. the himalayan mountains formed at a convergent plate boundary in this manner. the rocks are smashed together causing them to due to the intense heat and pressure from the colliding plates and eventually forming rock. a) melt; igneous b) layer; sedimentary c) recrystallize; metamorphic d) melt into the earth's interior; metamorphic
Answers: 1

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2
You know the right answer?
100 mL of a 0.300 M solution of AgNO3 reacts with 100 mL of a 0.300 M solution of HCl in a
coffee-c...
Questions



Social Studies, 12.02.2020 23:31


Business, 12.02.2020 23:31


Social Studies, 12.02.2020 23:31

Mathematics, 12.02.2020 23:31


Mathematics, 12.02.2020 23:31


Mathematics, 12.02.2020 23:31


Social Studies, 12.02.2020 23:31



Mathematics, 12.02.2020 23:31



Social Studies, 12.02.2020 23:31