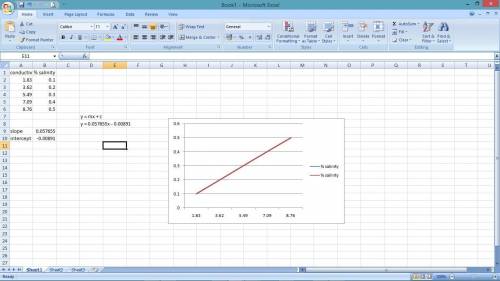
In chemistry lab student is determining the salinity of seawater using a conductivity meter. The student made standards of NaCl solutions and recorded the following data:
% Salinity Conductivity (ms)
0.10 1.83
0.20 3.62
0.30 5.49
0.40 7.09
0.50 8.76
The conductance of a 10 mL sample of unknown was found to be 5.82 ms.
Required:
a. Prepare a standard curve using the data above.
b. Determine the concentration of the unknown salt solution using the standard curve.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Complete the sentence. the lower the hydrogen ion concentration, the the ph. higher lower closer to 7 closer to 0
Answers: 2

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1
You know the right answer?
In chemistry lab student is determining the salinity of seawater using a conductivity meter. The stu...
Questions


Mathematics, 21.06.2019 23:30


Mathematics, 21.06.2019 23:30

Mathematics, 21.06.2019 23:30



Spanish, 21.06.2019 23:30



Mathematics, 21.06.2019 23:30


Mathematics, 21.06.2019 23:30


Mathematics, 21.06.2019 23:30