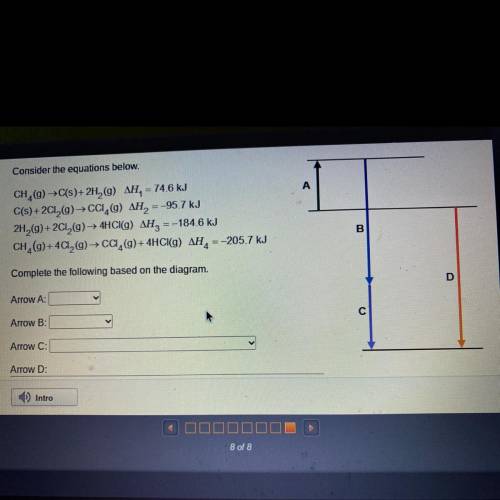
Consider the equations below.
A
CH (9) C(s)+2H (9) AH, = 74.6 kJ
C(s) +2012(9) ► CCI (9...

Consider the equations below.
A
CH (9) C(s)+2H (9) AH, = 74.6 kJ
C(s) +2012(9) ► CCI (9) AH, = -95.7 kJ
2H2(9)+2012(9) — 4HCl(9) AH2 =-184.6 kJ
CH (9)+ 4C12() → CC (9)+ 4HCI(g) AH, = -205.7 kJ
B
Complete the following based on the diagram.
D
Arrow A:
С
Arrow B:
Arrow C:
Arrow D:

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 15:00
Why does a plastic bottle that is sealed at a high altitude change it’s shape when taken to lower altitude
Answers: 2

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
Questions

Physics, 19.02.2020 20:23

History, 19.02.2020 20:23

Social Studies, 19.02.2020 20:23




Business, 19.02.2020 20:23






Mathematics, 19.02.2020 20:24


Mathematics, 19.02.2020 20:24








