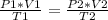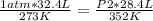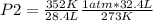Chemistry, 09.03.2021 02:50 lilbopeep21
A 32.4 L gas sample at STP is compressed to a volume of 28.4 L, and the temperature is increased to 352 K.
What is the new pressure of the gas in atmospheres? 1.47atm
How do I solve this?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 08:40
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2
You know the right answer?
A 32.4 L gas sample at STP is compressed to a volume of 28.4 L, and the temperature is increased to...
Questions



Mathematics, 14.02.2021 02:20

Mathematics, 14.02.2021 02:20

Engineering, 14.02.2021 02:20

Physics, 14.02.2021 02:20

Mathematics, 14.02.2021 02:20



Mathematics, 14.02.2021 02:20

Mathematics, 14.02.2021 02:20

Biology, 14.02.2021 02:20

Mathematics, 14.02.2021 02:20


Chemistry, 14.02.2021 02:20



Chemistry, 14.02.2021 02:30

Mathematics, 14.02.2021 02:30

History, 14.02.2021 02:30