
One essential step in the manufacture of many plant fertilizers is the production of nitrogen monoxide gas, NO(g). This step is represented by the following balanced equation: 4 NH3(g) + 5 O2(g) --> 4 NO(g) + 6 H2O(l) Determine the amount of oxygen needed to produce 1.2 x 104 mol of nitrogen monoxide gas.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2
You know the right answer?
One essential step in the manufacture of many plant fertilizers is the production of nitrogen monoxi...
Questions

SAT, 17.12.2020 01:00


Mathematics, 17.12.2020 01:00

Mathematics, 17.12.2020 01:00

English, 17.12.2020 01:00

Social Studies, 17.12.2020 01:00





Mathematics, 17.12.2020 01:00

Mathematics, 17.12.2020 01:00


Mathematics, 17.12.2020 01:00

Mathematics, 17.12.2020 01:00

Mathematics, 17.12.2020 01:00

Mathematics, 17.12.2020 01:00

Computers and Technology, 17.12.2020 01:00

Mathematics, 17.12.2020 01:00

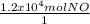 ×
× 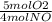 = 1.5 moles of O2 needed
= 1.5 moles of O2 needed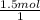 ×
× 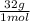 = 48 grams needed
= 48 grams needed

