
Chemistry, 10.03.2021 16:30 Albertrami9019
The tablet has a mass of 1.20 g and contains 700 mg of lithium carbonate. Calculate the percentage by mass of lithium carbonate in this tablet.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

You know the right answer?
The tablet has a mass of 1.20 g and contains 700 mg of lithium carbonate.
Calculate the percentage...
Questions

Social Studies, 08.01.2021 14:00


Mathematics, 08.01.2021 14:00

Chemistry, 08.01.2021 14:00


Mathematics, 08.01.2021 14:00

Social Studies, 08.01.2021 14:00

Social Studies, 08.01.2021 14:00

History, 08.01.2021 14:00



Mathematics, 08.01.2021 14:00


Health, 08.01.2021 14:00


Social Studies, 08.01.2021 14:00

Mathematics, 08.01.2021 14:00


History, 08.01.2021 14:00

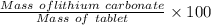

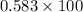
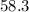 %
%

