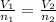Chemistry, 11.03.2021 04:20 dshood6599
If 5.50 mol of a gas occupies 230 L at constant temperature and pressure, how many moles are there if the volume is decreased to 176 L?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1


Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2
You know the right answer?
If 5.50 mol of a gas occupies 230 L at constant temperature and pressure, how many moles are there i...
Questions



Mathematics, 22.03.2021 18:00


Mathematics, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00



Computers and Technology, 22.03.2021 18:00




History, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00

Social Studies, 22.03.2021 18:00

Mathematics, 22.03.2021 18:00



Mathematics, 22.03.2021 18:00