
Chemistry, 11.03.2021 21:40 abdullahs4639
PLEASE WILL GIVE BRAINLIESTASAP
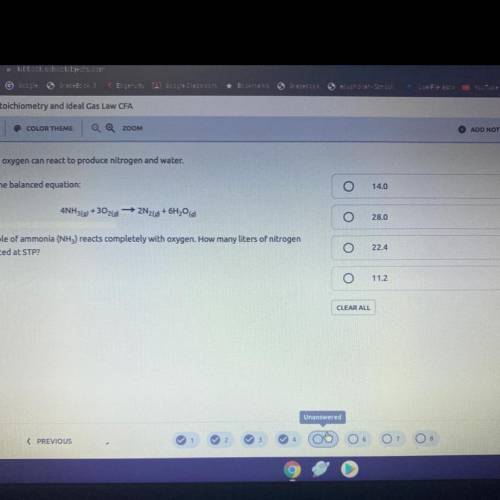
5. Ammonia and oxygen can react to produce nitrogen and water.
Following is the balanced equation:
4NH3(0)
+302(0)
- 2N269 + 6H20
Suppose 1 mole of ammonia (NH3) reacts completely with oxygen. How many liters of nitrogen
will be produced at STP?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 23.06.2019 15:00
What is the mass in grams of 0.94 moles of sodium bicarbonate, nahco3?
Answers: 1
You know the right answer?
PLEASE WILL GIVE BRAINLIESTASAP
5. Ammonia and oxygen can react to produce nitrogen and water.
Questions


English, 02.07.2019 19:50

Social Studies, 02.07.2019 19:50



Mathematics, 02.07.2019 19:50


History, 02.07.2019 19:50


Mathematics, 02.07.2019 19:50

History, 02.07.2019 19:50

Mathematics, 02.07.2019 19:50






Biology, 02.07.2019 19:50


Chemistry, 02.07.2019 19:50



