Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 21.06.2019 22:00
Which of the following statements is true about planck’s law
Answers: 1

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1
You know the right answer?
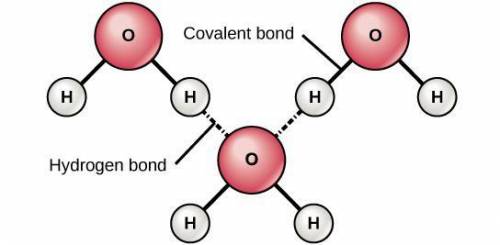
In a water molecule, what type of bond forms between the oxygen and hydrogen atoms...
Questions


English, 30.10.2020 21:40

Mathematics, 30.10.2020 21:40

Mathematics, 30.10.2020 21:40

Biology, 30.10.2020 21:40

Mathematics, 30.10.2020 21:40

Biology, 30.10.2020 21:40

Advanced Placement (AP), 30.10.2020 21:40



Computers and Technology, 30.10.2020 21:40


Medicine, 30.10.2020 21:40

English, 30.10.2020 21:40

Chemistry, 30.10.2020 21:40

Mathematics, 30.10.2020 21:40