
Chemistry, 18.03.2021 01:40 lisa123465
Select the correct processes. During which processes does the space between particles tend to increase?
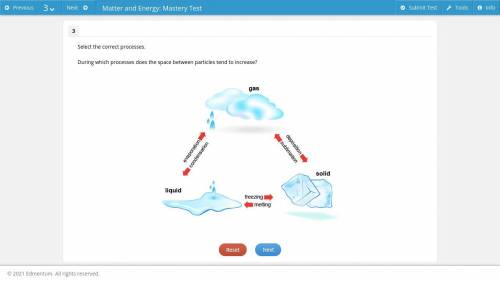

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3
You know the right answer?
Select the correct processes.
During which processes does the space between particles tend to incre...
Questions

English, 15.07.2019 06:10


Biology, 15.07.2019 06:10

History, 15.07.2019 06:10

Social Studies, 15.07.2019 06:10


Business, 15.07.2019 06:10

English, 15.07.2019 06:10


Chemistry, 15.07.2019 06:10

History, 15.07.2019 06:10

Social Studies, 15.07.2019 06:10

Social Studies, 15.07.2019 06:10


Business, 15.07.2019 06:10



History, 15.07.2019 06:10

Social Studies, 15.07.2019 06:10

Chemistry, 15.07.2019 06:10



