Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Nickel crystallizes in the face-centered cubic (fcc) lattice. the density of the metal is 8902 kg/m3. calculate the radius of a nickel atom.
Answers: 1

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 00:00
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 03:10
Between 2014 and 2016, more than 25,000 children in flint, michigan, drank water that was contaminated with lead from lead pipes. during this time, the city claimed the water was safe to drink. which of these actions could the city have taken to ensure that the drinking water was free from lead?
Answers: 3
You know the right answer?
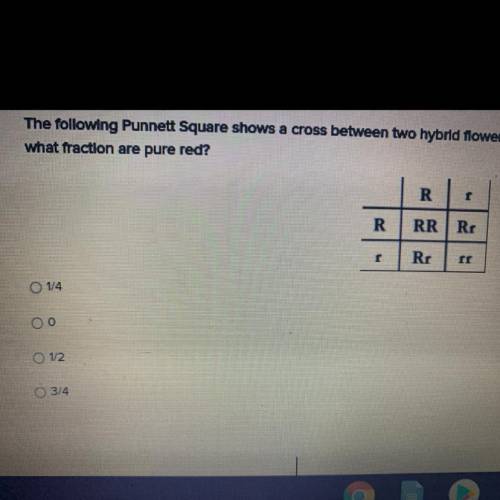
the following punnett square shows a cross between two hybrid flowers. if the flowers show incomplet...
Questions

Mathematics, 19.04.2021 19:30



Mathematics, 19.04.2021 19:30



Mathematics, 19.04.2021 19:30

Mathematics, 19.04.2021 19:30



Mathematics, 19.04.2021 19:30

Physics, 19.04.2021 19:30



Mathematics, 19.04.2021 19:30

Mathematics, 19.04.2021 19:30



Mathematics, 19.04.2021 19:30

Mathematics, 19.04.2021 19:30