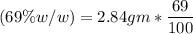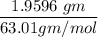Chemistry, 19.03.2021 18:10 rileybantaos1c8s
Mass of watch glass + filter paper = 105.98 g
Mass of watch glass + filter paper + crystallized product = 109.03 g
Mass of uncrystallized product (show work) =
Mass of methyl benzoate = 3.08 g
Volume of nitric acid used = 2.0 mL
Theoretical yield based on each of the starting materials
(Please use two Dimensional Analysis (DA) equations, one for the maximum amount of product obtainable from the amount of methyl benzoate you used and the other from the concentrated nitric acid, then use the lesser of the two to determine the Limiting Reagent; you must determine the number of moles in 2.00 mL of concentrated nitric acid [concentration 69.0% (w/w), and density (1.42 g/mL)].
Required:
a. Identity of the Limiting reagent (LR) based on the above two DA equations =
b. Max amount of product obtainable from the LR =
c. Mass of the product you obtained:

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
Mass of watch glass + filter paper = 105.98 g
Mass of watch glass + filter paper + crystallized pro...
Questions









Social Studies, 04.03.2021 21:20




Mathematics, 04.03.2021 21:20

Mathematics, 04.03.2021 21:20

Social Studies, 04.03.2021 21:20

Engineering, 04.03.2021 21:20

Mathematics, 04.03.2021 21:20

English, 04.03.2021 21:20

Chemistry, 04.03.2021 21:20

History, 04.03.2021 21:20