Which compound is more soluble in water at 25°C?
A.
MgF2 (Ksp = 5.2 x 10-11)
B.
S...

Chemistry, 19.03.2021 22:00 janiadavis35
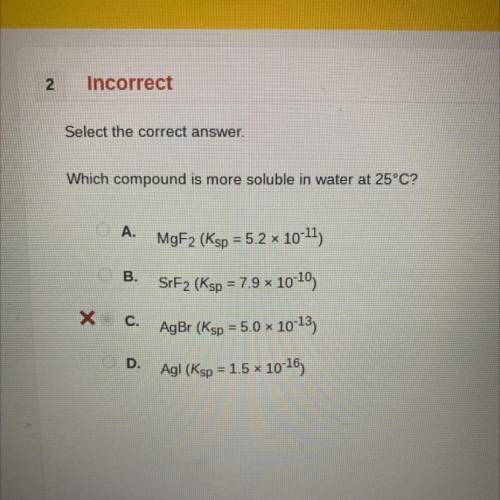
Which compound is more soluble in water at 25°C?
A.
MgF2 (Ksp = 5.2 x 10-11)
B.
SrF2 (Ksp = 7.9 x 10-10)
C.
AgBr (Ksp = 5.0 x 10-13)
D.
Agl (Ksp = 1.5 * 10-16)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3

You know the right answer?
Questions

Health, 19.07.2019 18:00


Mathematics, 19.07.2019 18:00

Physics, 19.07.2019 18:00



Physics, 19.07.2019 18:00

History, 19.07.2019 18:00

Mathematics, 19.07.2019 18:00











Chemistry, 19.07.2019 18:00