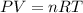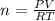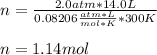Chemistry, 22.03.2021 05:50 AutumnGarringer
How many moles of ammonia gas can be formed from 14.0 L of hydrogen gas 300 K and a pressure of 2.0 atm

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
How many moles of ammonia gas can be formed from 14.0 L of hydrogen gas 300 K and a pressure of 2.0...
Questions


Social Studies, 26.02.2022 06:00

Mathematics, 26.02.2022 06:00





Mathematics, 26.02.2022 06:00

Business, 26.02.2022 06:00

Chemistry, 26.02.2022 06:10






Biology, 26.02.2022 06:10


History, 26.02.2022 06:10