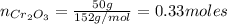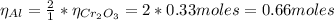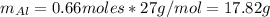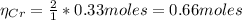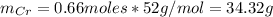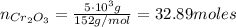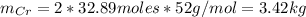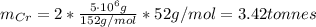Chromium is manufactured by heating a mixture of chromium(III) oxide with aluminium powder.
Cr2O3(s) + 2Al(s) → 2Cr(s) + Al2O3(s)
a=Calculate the mass of aluminium needed to react with 50 g of Cr2O3.
b=Calculate the mass of chromium produced from 50 g of Cr2O3.
c=Calculate the mass of chromium produced from 5 kg of Cr2O3.
d=Calculate the mass of chromium produced from5 tonnes of Cr2O3.
Note: 1 tonne = 1,000,000 g
Ar: Cr = 52, O = 16, Al = 27

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3

Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3

Chemistry, 23.06.2019 05:30
Suppose you discovered a new element with 120 protons and 2 electrons in its outer level . i'm what group does this new element belong? what properties would you expect it to have
Answers: 1
You know the right answer?
Chromium is manufactured by heating a mixture of chromium(III) oxide with aluminium powder.
Cr2O3(s...
Questions


Mathematics, 27.09.2020 17:01




Mathematics, 27.09.2020 17:01

History, 27.09.2020 17:01

Mathematics, 27.09.2020 17:01

Biology, 27.09.2020 17:01



Computers and Technology, 27.09.2020 17:01

Social Studies, 27.09.2020 17:01






History, 27.09.2020 17:01

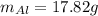
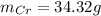
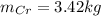
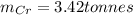
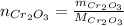
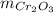 : is the mass = 50 g
: is the mass = 50 g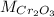 : is the molar mass = 2*52+3*16 = 152 g/mol
: is the molar mass = 2*52+3*16 = 152 g/mol