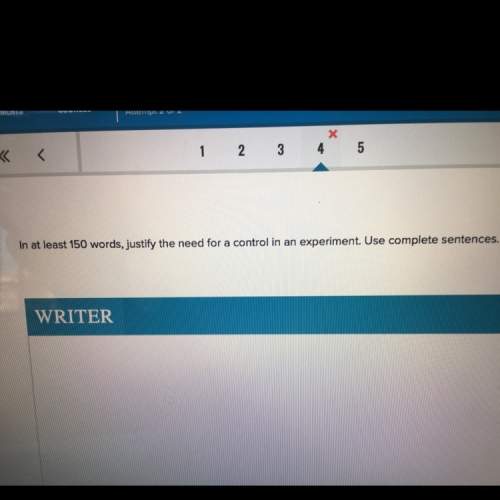
Chemistry, 22.03.2021 22:00 gujaratif932
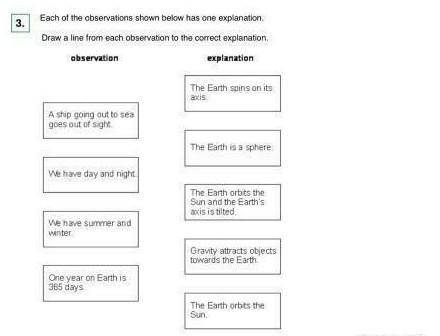
HELPP ILL GUVE YOU LOTS OF POINTS EVERYTHING U WANT I REALLY NEED HELP

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
HELPP ILL GUVE YOU LOTS OF POINTS EVERYTHING U WANT I REALLY NEED HELP
...
...
Questions

Mathematics, 15.04.2021 19:40

English, 15.04.2021 19:40

Mathematics, 15.04.2021 19:40

Biology, 15.04.2021 19:40

Mathematics, 15.04.2021 19:40






Mathematics, 15.04.2021 19:40

Spanish, 15.04.2021 19:40








History, 15.04.2021 19:40