PLEASE HELPP!
The proposed mechanism for a reaction is:
Step 1: A + B X (fast)
Step 2:...

Chemistry, 24.03.2021 22:40 nnaaatt1845
PLEASE HELPP!
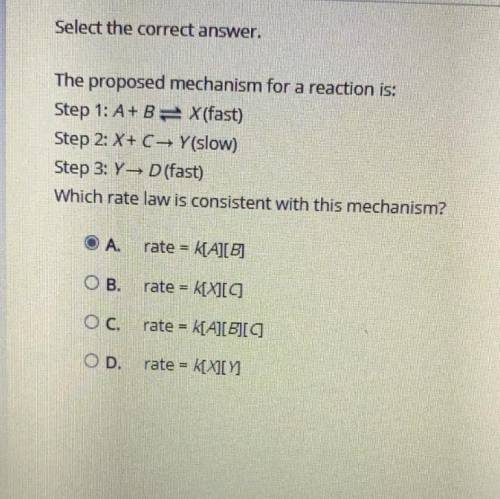
The proposed mechanism for a reaction is:
Step 1: A + B X (fast)
Step 2: X + C → Y (slow)
Step 3: Y → D (fast)
Which rate law is consistent with this mechanism?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Subduction zones occur on earth where dense oceanic crust dives under more buoyant continental crust. these boundaries are characterized by a deep ocean trench next to a high continental mountain range, large numbers of earthquakes and volcanoes. all of this is further evidence for the a) big bang theory b) origin of the species eliminate c theory of plate tectonics d theory of natural selection 4 sedimentary rocks found high in the himalayen mountain
Answers: 1

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
Questions

Mathematics, 29.03.2021 01:00



Physics, 29.03.2021 01:00




Mathematics, 29.03.2021 01:00


Mathematics, 29.03.2021 01:00

Mathematics, 29.03.2021 01:00

Chemistry, 29.03.2021 01:00

Advanced Placement (AP), 29.03.2021 01:00

Mathematics, 29.03.2021 01:00

Mathematics, 29.03.2021 01:00




English, 29.03.2021 01:00



