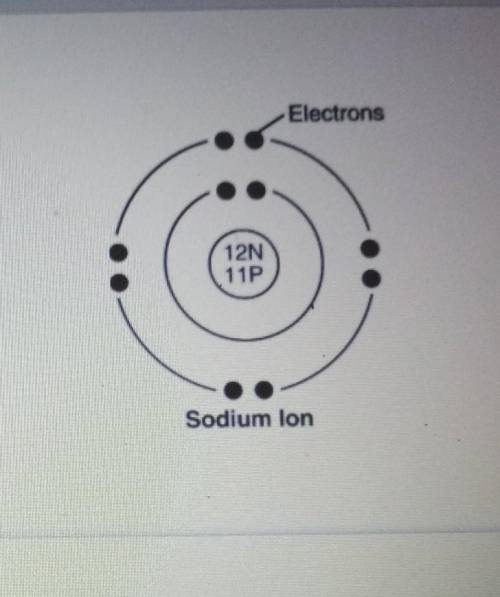
What is the electrical charge of this ion
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 14:30
If energy was included in a chemical reaction, on which side of the equation would it be written for an endothermic reaction?
Answers: 1
You know the right answer?
Questions

Mathematics, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57


History, 04.06.2020 06:57


Mathematics, 04.06.2020 06:57

English, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Business, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57


Biology, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57

Chemistry, 04.06.2020 06:57

Mathematics, 04.06.2020 06:57