
Chemistry, 26.03.2021 01:00 kaliyab191
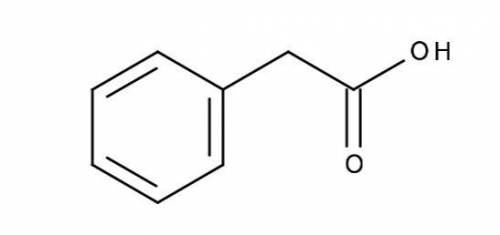
Calculate (a) the pH of a 0.0250 M solution of phenylacetic acid, and (b) the pH of a 0.0500 M solution of sodium phenylacetate. The pKa of phenylacetic acid is 4.31, and its structure is shown below.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1
You know the right answer?
Calculate (a) the pH of a 0.0250 M solution of phenylacetic acid, and (b) the pH of a 0.0500 M solut...
Questions

Mathematics, 16.04.2020 18:30

Mathematics, 16.04.2020 18:30

Social Studies, 16.04.2020 18:30




Computers and Technology, 16.04.2020 18:30

English, 16.04.2020 18:30


English, 16.04.2020 18:30

English, 16.04.2020 18:30

Mathematics, 16.04.2020 18:30

English, 16.04.2020 18:30




English, 16.04.2020 18:30





