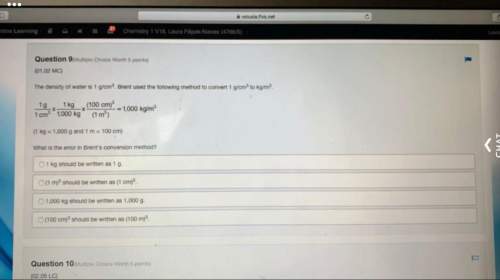
Limiting Reactants: on a video my teacher made, he said
4 H2 + 100 O2 = 4 H2O
However,...

Limiting Reactants: on a video my teacher made, he said
4 H2 + 100 O2 = 4 H2O
However, later he said,
15 H2 + 12 O2 = 7 H2O
Wouldn’t it be 15 H2O because H2O consists of H2 and a single O atom? In the equation there are 15 H2 and 24 single O atoms. Did my teacher make an error, or did I?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 22.06.2019 03:30
Nanotechnology, the field of trying to build ultrasmall structures one atom at a time, has progressed in recent years. one potential application of nanotechnology is the construction of artificial cells. the simplest cells would probably mimic red blood cells, the body's oxygen transporters. for example, nanocontainers, perhaps constructed of carbon, could be pumped full of oxygen and injected into a person's bloodstream. if the person needed additional oxygen-due to a heart attack perhaps, or for the purpose of space travel-these containers could slowly release oxygen into the blood, allowing tissues that would otherwise die to remain alive. suppose that the nanocontainers were cubic and had an edge length of 24 nanometers. part a part complete what is the volume of one nanocontainer? (ignore the thickness of the nanocontainer's wall.) express your answer using two significant figures. v v = 1.4ă—10â’20 l previous answers correct significant figures feedback: your answer 1.3824â‹…10â’20 = 1.382ă—10â’20 l was either rounded differently or used a different number of significant figures than required for this part. if you need this result for any later calculation in this item, keep all the digits and round as the final step before submitting your answer. part b suppose that each nanocontainer could contain pure oxygen pressurized to a density of 81 g/l . how many grams of oxygen could be contained by each nanocontainer?
Answers: 3


Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1
You know the right answer?
Questions

Social Studies, 31.08.2019 07:30

Mathematics, 31.08.2019 07:30

Social Studies, 31.08.2019 07:30

Social Studies, 31.08.2019 07:30


History, 31.08.2019 07:30

History, 31.08.2019 07:30

Biology, 31.08.2019 07:30

English, 31.08.2019 07:30


Computers and Technology, 31.08.2019 07:30


Mathematics, 31.08.2019 07:30






Mathematics, 31.08.2019 07:30

Biology, 31.08.2019 07:30