

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 22.06.2019 21:30
In one or two grammatically correct sentences, write a definition for the term molecule geometry
Answers: 3
You know the right answer?
What volume of oxygen gas is released at stp if 10.0 g of potassium chlorate is decomposed? (the mo...
Questions


History, 09.10.2019 03:40


Mathematics, 09.10.2019 03:40


History, 09.10.2019 03:40


Arts, 09.10.2019 03:40


English, 09.10.2019 03:40


History, 09.10.2019 03:40


Spanish, 09.10.2019 03:40

History, 09.10.2019 03:40

Spanish, 09.10.2019 03:40

Computers and Technology, 09.10.2019 03:40

History, 09.10.2019 03:40


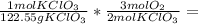 0.1224 mol O₂
0.1224 mol O₂

