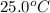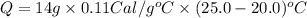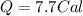Chemistry, 16.10.2019 15:30 oofoofoof1
Suppose a 14-gram sample of iron is heated from 20.0°c to 25.0°c. the specific heat of iron is 0.11 cal/g°c. how much heat energy was absorbed by the iron? 38.5 cal 7.7 cal 636 cal 69.3 cal

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1
You know the right answer?
Suppose a 14-gram sample of iron is heated from 20.0°c to 25.0°c. the specific heat of iron is 0.11...
Questions






English, 31.12.2020 14:00

Mathematics, 31.12.2020 14:00


Social Studies, 31.12.2020 14:00




History, 31.12.2020 14:00

Mathematics, 31.12.2020 14:00

History, 31.12.2020 14:00




Mathematics, 31.12.2020 14:00

Social Studies, 31.12.2020 14:00

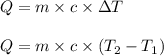
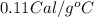
 = change in temperature
= change in temperature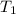 = initial temperature =
= initial temperature = 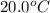
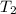 = final temperature =
= final temperature =