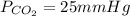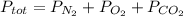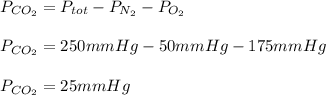Chemistry, 26.03.2021 19:40 Damagingawsomeness2
A gas mixture contains oxygen, nitrogen, and carbon dioxide. It has a pressure of 250 mmHg. If the pressure of oxygen is 50 mmHg and the pressure of nitrogen is 175 mmHg then what is the pressure for carbon dioxide?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:50
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
A gas mixture contains oxygen, nitrogen, and carbon dioxide. It has a pressure of 250 mmHg. If the p...
Questions


Mathematics, 06.11.2020 09:30

Chemistry, 06.11.2020 09:30

Mathematics, 06.11.2020 09:30






Chemistry, 06.11.2020 09:30

Mathematics, 06.11.2020 09:30


Mathematics, 06.11.2020 09:30

Mathematics, 06.11.2020 09:30

Law, 06.11.2020 09:30



Advanced Placement (AP), 06.11.2020 09:30

English, 06.11.2020 09:30

Engineering, 06.11.2020 09:30