
Chemistry, 29.03.2021 19:10 kendramiller3965
A saturated solution of KClO3 is formed from 100 g of water. If the saturated solution is quickly cooled from 90∘C to 50∘C, how many grams of precipitate are formed?
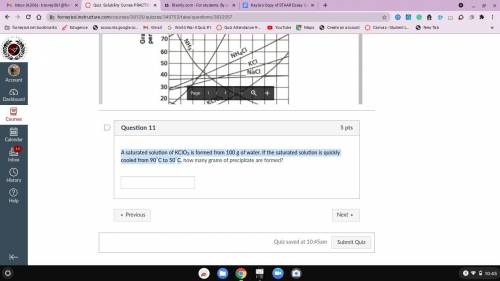

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:40
It is important to wear proper protective equipment in lab even when not actively performing experiments because accidents can affect any researcher, even one not working on an experiment. select the best answer from the choices provided
Answers: 3

Chemistry, 21.06.2019 22:00
Bohr's model could only explain the spectra of which type of atoms? single atoms with one electron single atoms with more than one electron bonded atoms with one electron bonded atoms with more than one electron
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2
You know the right answer?
A saturated solution of KClO3 is formed from 100 g of water. If the saturated solution is quickly co...
Questions





Health, 28.08.2019 14:00

Chemistry, 28.08.2019 14:00


Biology, 28.08.2019 14:00

Arts, 28.08.2019 14:00

Mathematics, 28.08.2019 14:00

History, 28.08.2019 14:00

Mathematics, 28.08.2019 14:00



Mathematics, 28.08.2019 14:00


Biology, 28.08.2019 14:00



Biology, 28.08.2019 14:00



