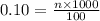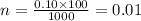Chemistry, 29.03.2021 23:40 Janznznz4012
Molarity and Dilution How many moles are needed in a 100. mL solution of a 0.10M CuSO4 5H2O solution?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1
You know the right answer?
Molarity and Dilution
How many moles are needed in a 100. mL solution of a 0.10M CuSO4 5H2O solutio...
Questions







Biology, 10.07.2019 03:00

Chemistry, 10.07.2019 03:00



Mathematics, 10.07.2019 03:00


Mathematics, 10.07.2019 03:00

History, 10.07.2019 03:00

History, 10.07.2019 03:00

History, 10.07.2019 03:00

History, 10.07.2019 03:00

History, 10.07.2019 03:00

History, 10.07.2019 03:00

Social Studies, 10.07.2019 03:00

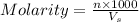
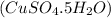 = ?
= ?  = volume of solution in ml
= volume of solution in ml