
Chemistry, 30.03.2021 01:00 kamkam1713
A gas tank contains 2.5 moles of oxygen, 7.0 moles of nitrogen and 0.5 mole of water vapor. The total pressure of the mixture is 852 torr. Calculate the partial pressure of each gas. Show work.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 23.06.2019 00:10
Apropane torch is lit inside a hot air balloon during preflight preparations to inflate the balloon. which condition of the gas remains constant
Answers: 2
You know the right answer?
A gas tank contains 2.5 moles of oxygen, 7.0 moles of nitrogen and 0.5 mole of water vapor. The tota...
Questions


English, 23.11.2020 14:00





English, 23.11.2020 14:00



Mathematics, 23.11.2020 14:00


Chemistry, 23.11.2020 14:00

English, 23.11.2020 14:00

Mathematics, 23.11.2020 14:00




Mathematics, 23.11.2020 14:00


Arts, 23.11.2020 14:00

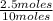 = 0.25mole fraction of nitrogen =
= 0.25mole fraction of nitrogen = 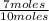 = 0.7mole fraction of water vapor =
= 0.7mole fraction of water vapor = 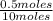 = 0.05
= 0.05

