
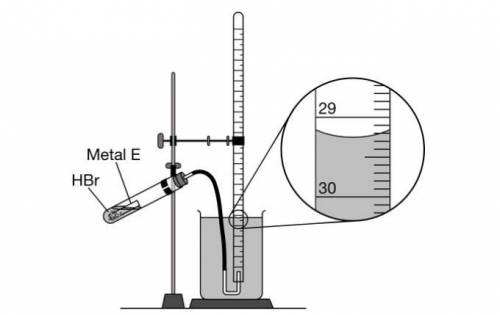
As shown in the following diagram, 10 mL samples of HBr(aq) are added to different quantities of E(s) in a closed test tube. At the start of each trial, the gas-collecting tube is filled completely with distilled water. The samples of E(s) react with HBr(aq) , and the H2(g) produced is collected in the gas-collecting tube.
The enlarged view of the gas-collecting tube at the end of trial 3 is shown in the diagram.
What should the student record as the volume, in mL , of H2(g) collected in trial 3?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 14:20
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d.lytic
Answers: 1

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 19:50
What is the wavelength of a wave with a velocity of 50 m/s and a frequency of 5hz a 250 m b 0.1 m c 10m d 0.01 m
Answers: 2
You know the right answer?
As shown in the following diagram, 10 mL samples of HBr(aq) are added to different quantities of E(s...
Questions


Geography, 24.02.2021 04:30

English, 24.02.2021 04:30

Mathematics, 24.02.2021 04:30





English, 24.02.2021 04:30

Mathematics, 24.02.2021 04:40

Mathematics, 24.02.2021 04:40



Health, 24.02.2021 04:40

Chemistry, 24.02.2021 04:40

Mathematics, 24.02.2021 04:40






