ANSWER NEEDED ASAP!!
How many grams of Cl2 {molar mass = 70.9 g/mol} gas can be produced
when...

Chemistry, 31.03.2021 19:50 NearNoodle23
ANSWER NEEDED ASAP!!
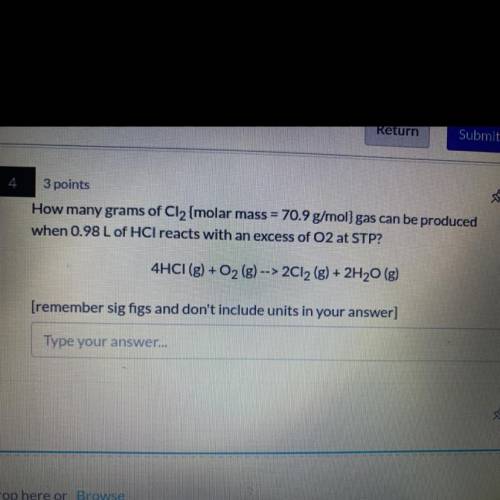
How many grams of Cl2 {molar mass = 70.9 g/mol} gas can be produced
when 0.98 L of HCl reacts with an excess of O2 at STP?
4HCI (g) + O2 (g) --> 2012 (g) + 2H20 (g)
[remember sig figs and don't include units in your answer]

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
List the two type of transporst that the cell in orde to transport molecules acroos the membrane
Answers: 1


Chemistry, 22.06.2019 18:00
Alidded glass container is filled with a colored gas. after a period of time, it is observed that the gas is uniformly spread throughout the box and that the movement has slowed considerably. next, a warm iron plate is carefully placed under the box. why is there resumed movement of the gas in the container?
Answers: 2

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1
You know the right answer?
Questions

Mathematics, 07.01.2021 06:10



Biology, 07.01.2021 06:10



Mathematics, 07.01.2021 06:10

Mathematics, 07.01.2021 06:10


Biology, 07.01.2021 06:10



Mathematics, 07.01.2021 06:10

Mathematics, 07.01.2021 06:10





Biology, 07.01.2021 06:10

Spanish, 07.01.2021 06:10



