Chemistry, 01.04.2021 21:50 nataliahenderso
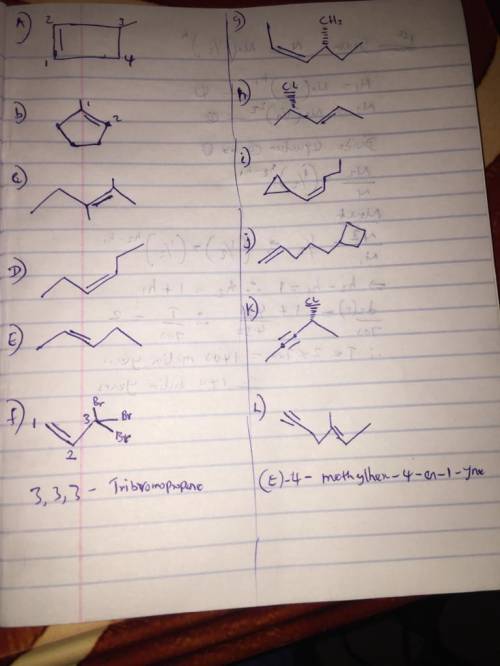
Write a structural formula for each of the following: (a) 3-Methylcyclobutene (b) 1-Methylcyclopentene (c) 2,3-Dimethyl-2-pentene (d) (Z)-3-Hexene (e) (E)-2-Pentene (f) 3,3,3-Tribromopropene (g) (Z,4R)-4-Methyl-2-hexene (h) (E,4S )-4-Chloro-2-pentene (i) (Z)-1-Cyclopropyl-1-pentene (j) 5-Cyclobutyl-1-pentene (k) (R)-4-Chloro-2-pentyne (l) (E)-4-Methylhex-4-en-1-yne

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1
You know the right answer?
Write a structural formula for each of the following: (a) 3-Methylcyclobutene (b) 1-Methylcyclopente...
Questions


Geography, 24.08.2019 13:10

Social Studies, 24.08.2019 13:10


History, 24.08.2019 13:10



History, 24.08.2019 13:10

Chemistry, 24.08.2019 13:10


Mathematics, 24.08.2019 13:10

Mathematics, 24.08.2019 13:10






Mathematics, 24.08.2019 13:10

Health, 24.08.2019 13:10