
Chemistry, 02.04.2021 01:20 anthonylopez1
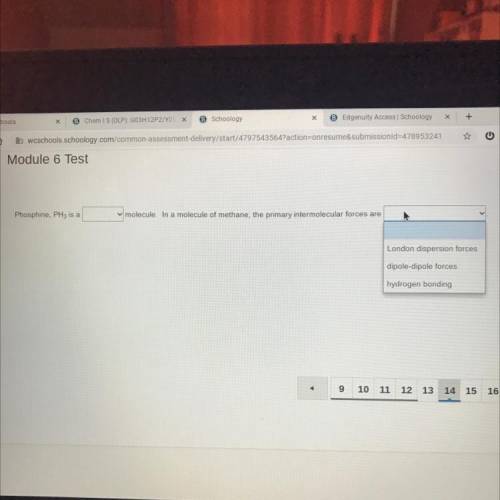
Phosphine, PH3 is a (polar, nonpolar) molecule. In a molecule of methane, the primary intermolecular forces are (london dispersion forces, dipole-dipole forces, hydrogen bonding)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 23.06.2019 10:10
Solid tin exists in two forms: white and gray. for the transformation sn(s, white) → sn(s, gray) the enthalpy change is -2.1 kj/mol and the entropy change is -7.4 j/(mol*k). a. calculate the gibbs free energy change for the conversion of 1.00 mol white tin to gray tin at -30℃. b. will white tin convert spontaneously to gray tin at -30℃? c. at what temperature are white and gray tin thermodynamically equivalent at a pressure of 1 atm?
Answers: 3
You know the right answer?
Phosphine, PH3 is a (polar, nonpolar) molecule. In a molecule of methane, the primary intermolecular...
Questions



Advanced Placement (AP), 07.07.2019 12:00

English, 07.07.2019 12:00



Mathematics, 07.07.2019 12:00

English, 07.07.2019 12:00





Mathematics, 07.07.2019 12:00

Chemistry, 07.07.2019 12:00


Mathematics, 07.07.2019 12:00

Mathematics, 07.07.2019 12:00

Mathematics, 07.07.2019 12:00




