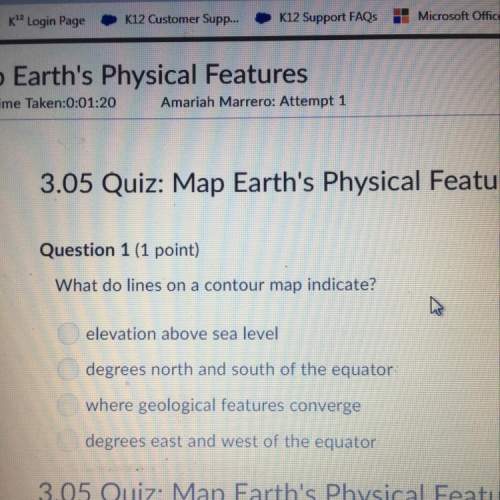
Chemistry, 02.04.2021 05:20 Elijah7934
What is the oxidation state of the underlined atom in the reaction:
Zn + 2HCl --> ZnCl2 + H2
underlined is H in 2HCl

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

You know the right answer?
What is the oxidation state of the underlined atom in the reaction:
Zn + 2HCl --> ZnCl2 + H2
Questions

English, 22.01.2020 23:31











Business, 22.01.2020 23:31


Physics, 22.01.2020 23:31