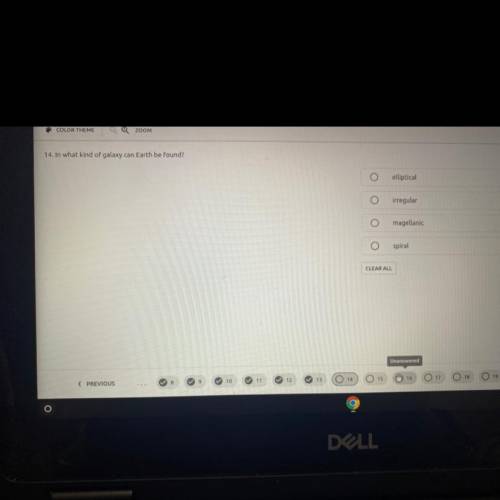
Please please help :/
...

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1
You know the right answer?
Questions

Mathematics, 17.06.2020 12:57

English, 17.06.2020 12:57

History, 17.06.2020 12:57


Mathematics, 17.06.2020 12:57

English, 17.06.2020 12:57

Health, 17.06.2020 12:57


Mathematics, 17.06.2020 13:57

History, 17.06.2020 13:57


Engineering, 17.06.2020 13:57

Chemistry, 17.06.2020 13:57


English, 17.06.2020 13:57

Mathematics, 17.06.2020 13:57

Health, 17.06.2020 13:57

Biology, 17.06.2020 13:57

History, 17.06.2020 13:57