
Chemistry, 02.04.2021 08:10 smithmorgan773p35885
If 10.00 moles of copper are reacted with 6.00 moles of sulfur according to the following balanced equation, which reactant is the limiter and how many moles of excess reactant would remain after the reaction is completed? 2 Cu + S --> Cu2S
Cu is limiting and 1.00 mole of excess S remain
S is limiting and 1.00 moles of excess Cu remain
S is limiting and 4.00 moles of excess Cu remain
Cu is limiting and 4.00 moles of excess S remain

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which of the following pairs of elements belong to the same groupa. h and he b. li and bec. c and pb d. ga and ge
Answers: 1

Chemistry, 21.06.2019 23:00
What is the volume of the fluid in the graduated cylinder with accuracy and measured to the correct degree of precision? 41.2 ml 42.0 ml 41.23 ml 41.89 ml
Answers: 1


You know the right answer?
If 10.00 moles of copper are reacted with 6.00 moles of sulfur according to the following balanced e...
Questions


History, 28.07.2019 17:00




Mathematics, 28.07.2019 17:00






Physics, 28.07.2019 17:00



English, 28.07.2019 17:00



English, 28.07.2019 17:00

Mathematics, 28.07.2019 17:00

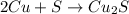
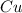 require 1 mole of
require 1 mole of 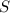
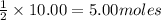 of
of 

