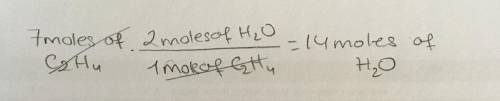Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 23.06.2019 12:30
15) a substance used in manufacturing gasoline consists of finely divided platinum supported on an inert solid. suppose that the platinum is formed by the high temperature reaction between platinum (iv) oxide and hydrogen gas. the other product is water. a) write and balance the equation b) how many grams of hydrogen are needed to produce 1.0 g of platinum metal? c) how many moles of water are produced at the same time? how many grams? ( show work, .)
Answers: 1

Chemistry, 23.06.2019 13:30
Malik formed a hypothesis that an increase in atmospheric oxygen levels by 10% would cause red-legged grasshoppers to grow larger than normal. suppose that malik performs an experiment to test his hypothesis. which of these actions would represent a scientific mistake in his experiment? a. he experiments on live grasshoppers instead of preserved ones. b. he focuses on red-legged grasshoppers instead of all kinds of grasshoppers. c. he varies the nitrogen and carbon dioxide levels in the air from one trial to the next. d. he conducts the experiment in a controlled lab setting with a lab partner. e. he measures the mass and length of his specimens at the start of each trial.
Answers: 1
You know the right answer?
How many grams of H2O would be produced if you started with 7 moles of ethylene (C2H4 )?...
Questions


History, 01.12.2020 23:00

Mathematics, 01.12.2020 23:00

Mathematics, 01.12.2020 23:00

Mathematics, 01.12.2020 23:00

Mathematics, 01.12.2020 23:00

World Languages, 01.12.2020 23:00






History, 01.12.2020 23:00


Mathematics, 01.12.2020 23:00

English, 01.12.2020 23:00

Mathematics, 01.12.2020 23:00


Mathematics, 01.12.2020 23:00

History, 01.12.2020 23:00