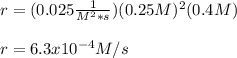Study the reaction and read the statement.
Xe+3F2→XeF6
The rate constant for this reaction is...

Study the reaction and read the statement.
Xe+3F2→XeF6
The rate constant for this reaction is 0.025, and the reaction is second order in Xe and first order in F2.
What is the rate of the reaction if [Xe] is 0.25 M, and [F2] is 0.4 M?
4.0 × 10–^4
2.5 × 10–^3
1.5 × 10–^3
6.3 × 10–^4

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration?
Answers: 3

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 23:40
What energy conversion occurs when a sling shot is used to shoot a rock across the room? (2 points) question 2 options: 1) stored mechanical energy is converted to mechanical energy. 2) stored mechanical energy is converted to radiant energy. 3) gravitational energy is converted to radiant energy. 4) gravitational energy is converted to mechanical energy.
Answers: 1
You know the right answer?
Questions

History, 11.09.2019 22:20


Social Studies, 11.09.2019 22:20


Mathematics, 11.09.2019 22:20






Advanced Placement (AP), 11.09.2019 22:20









![r=k[Xe]^2[F_2]](/tpl/images/1238/0471/c5483.png)