
Chemistry, 05.04.2021 02:40 dhhdjejdjdbdhdne
You have 2344.44 grams of water (H20) that react according to this balanced
chemical reaction. What is the theoretical yield of H3PO4 ?
P4010 + 6H2O --> 4H3PO4

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1


Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1
You know the right answer?
You have 2344.44 grams of water (H20) that react according to this balanced
chemical reaction. What...
Questions




Biology, 07.11.2019 06:31



Engineering, 07.11.2019 06:31

Biology, 07.11.2019 06:31

History, 07.11.2019 06:31



Social Studies, 07.11.2019 06:31


Mathematics, 07.11.2019 06:31




Biology, 07.11.2019 06:31


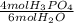 = 86.8 mol H₃PO₄
= 86.8 mol H₃PO₄

