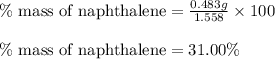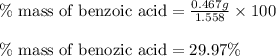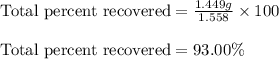Chemistry, 06.04.2021 05:00 aileenf598
Data Collection
Mass of the original sample of mixture (g) 1.558
Mass of recovered naphthalene (g) 0.483
Mass of recovered 3-nitroaniline (g) 0.499
Mass of recovered benzoic acid (g) 0.467
Calculations:
a. % by mass of naphthalene in original sample.
b. % by mass of 3-nitroaniline in original sample.
c. % by mass of benzoic acid in original sample.
d. total percent recovered.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2
You know the right answer?
Data Collection
Mass of the original sample of mixture (g) 1.558
Mass of recovered naphthale...
Mass of recovered naphthale...
Questions




Mathematics, 10.05.2021 18:20

History, 10.05.2021 18:20

Mathematics, 10.05.2021 18:20


English, 10.05.2021 18:20

English, 10.05.2021 18:20




SAT, 10.05.2021 18:20

History, 10.05.2021 18:20

French, 10.05.2021 18:20

Mathematics, 10.05.2021 18:20

Mathematics, 10.05.2021 18:20



Mathematics, 10.05.2021 18:20

 ......(1)
......(1)