

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1

You know the right answer?
A buffer is made with 0.493 M of the weak base methylamine () with 0.493 M (KA = 5.6 x 10^-4)
What...
Questions

Mathematics, 07.05.2021 07:10

Social Studies, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10

Mathematics, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10



Mathematics, 07.05.2021 07:10


Mathematics, 07.05.2021 07:10

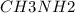 ) with 0.493 M
) with 0.493 M 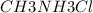 (KA = 5.6 x 10^-4)
What is the pH of this buffer system?
(KA = 5.6 x 10^-4)
What is the pH of this buffer system?

