
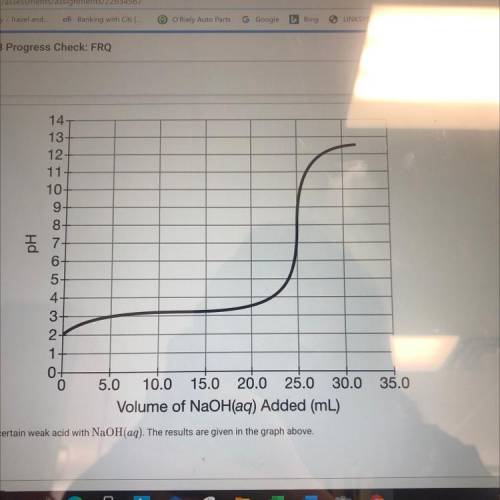
(C) a student ti trates 50.0mL of a 0.1 M solution of a certain weak acid with NaOH(aq). The results are given in the graph above (ii) what specific value from the graph is needed, in addition to the information in part (c) above, to calculate the molar concentration of the NaOH(aq)?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 22:00
11) burning your hand when accidentally touching a hot plate is an example of which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 2

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2

Chemistry, 23.06.2019 14:20
What kind of chemical reaction does the chemical equation sodium + chlorine → sodium chloride represent? a. combustion b. decomposition c. single replacement d. synthesis
Answers: 1

Chemistry, 24.06.2019 01:40
3. mr. hill has 27 students in his classand mr. chang has 24 students in hisclass. both classes will be divided intoequal sized teams within their ownclasses. what is the greatest number ofstudents that can be on team so that allthe teams are an equal number?
Answers: 2
You know the right answer?
(C) a student ti trates 50.0mL of a 0.1 M solution of a certain weak acid with NaOH(aq). The results...
Questions



Mathematics, 10.07.2019 18:00

Biology, 10.07.2019 18:00

Mathematics, 10.07.2019 18:00


Mathematics, 10.07.2019 18:00

Chemistry, 10.07.2019 18:00

Mathematics, 10.07.2019 18:00

English, 10.07.2019 18:00

Mathematics, 10.07.2019 18:00

Mathematics, 10.07.2019 18:00


Mathematics, 10.07.2019 18:00

Physics, 10.07.2019 18:00

History, 10.07.2019 18:00

Mathematics, 10.07.2019 18:00



Mathematics, 10.07.2019 18:00



