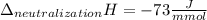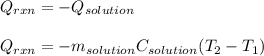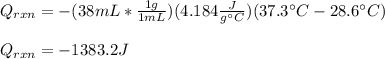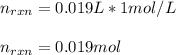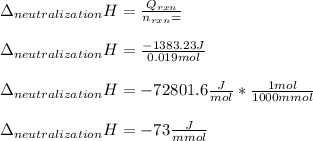Determine the enthalpy of neutralization in Joules/mmol for a solution resulting from 19 mL of 1 M NaOH solution and 19 mL of a HCl with the same molarity. If separately, each had a temperature of 28.6 degrees Celsius, and upon addition, the highest temperature reached by the solution was graphically determined to be 37.3 degrees Celsius. Round to the nearest whole number.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
Determine the enthalpy of neutralization in Joules/mmol for a solution resulting from 19 mL of 1 M N...
Questions

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01


English, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01


Mathematics, 09.09.2020 14:01

Computers and Technology, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01

Mathematics, 09.09.2020 14:01