
Chemistry, 08.04.2021 22:10 mariap3504
Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of MgO for each trial.
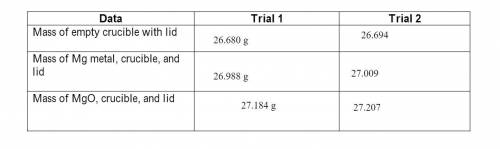

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:02
What is the partial pressure of ethane, pethane, in the flask?
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1
You know the right answer?
Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of MgO for ea...
Questions

Mathematics, 22.02.2021 21:40


Arts, 22.02.2021 21:40





Mathematics, 22.02.2021 21:40






Business, 22.02.2021 21:40



Mathematics, 22.02.2021 21:40

Chemistry, 22.02.2021 21:40

Mathematics, 22.02.2021 21:40



