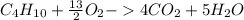Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:00
This graph gives information on changes in sea ice extent in the arctic ocean over a 30-year span. the overall trend shows in the ice extent. to address the trend, scientists need to ask themselves, one direct consequence of the trend is that
Answers: 1

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2
You know the right answer?
How many moles of carbon dioxide are produced when 0.25 mole of butane is consumed?...
Questions





Arts, 24.06.2019 16:00


Biology, 24.06.2019 16:00

Geography, 24.06.2019 16:00

History, 24.06.2019 16:00


German, 24.06.2019 16:00

Biology, 24.06.2019 16:00





History, 24.06.2019 16:00

Mathematics, 24.06.2019 16:00

Advanced Placement (AP), 24.06.2019 16:00

Biology, 24.06.2019 16:00