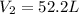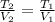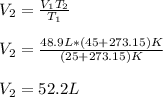Chemistry, 11.04.2021 05:00 madiforkner
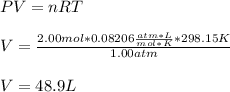
A gas system contains 2.00 moles of O2 and CO2 gas, has an initial temperature of 25.0 oC and is under 1.00 atm of pressure. If the pressure remains constant and the temperature is raised to 45.0 oC, then what will the new volume (assuming a closed system) be?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1
You know the right answer?
A gas system contains 2.00 moles of O2 and CO2 gas, has an initial temperature of 25.0 oC and is und...
Questions

Arts, 18.08.2021 23:10




Mathematics, 18.08.2021 23:10


English, 18.08.2021 23:10


Social Studies, 18.08.2021 23:10

History, 18.08.2021 23:10






Social Studies, 18.08.2021 23:10


Mathematics, 18.08.2021 23:10


English, 18.08.2021 23:10