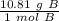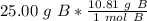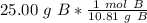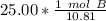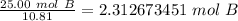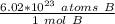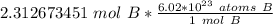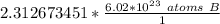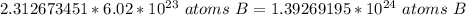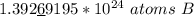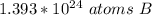1.333 x 10^23 atoms of B

Chemistry, 11.04.2021 05:50 giselabarajas24
How many atoms are in 25.00 g of B.
1.393 x 10^24 atoms of B
1.333 x 10^23 atoms of B
2.16 x 10^26 atoms of B
2.17 x 10^24 atoms of B

Answers: 3


Another question on Chemistry

Chemistry, 23.06.2019 09:00
How many moles of sulfur dioxide are in 2.26 × 10^33 sulfur dioxide molecules?
Answers: 3

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1

Chemistry, 23.06.2019 10:30
Fill in the blanks for the following statements: the rms speed of the molecules in a sample of h2 gas at 300 k will be times larger than the rms speed of o2 molecules at the same temperature, and the ratio µrms (h2) / µrms (o2) with increasing temperature. a not enough information is given to answer this question b sixteen, will not change c four, will not change d four, will increase e sixteen, will decrease
Answers: 2

Chemistry, 23.06.2019 14:00
Cassandra made a venn diagram to compare and contrast the two stages of cellular respiration. which belongs in the area marked x? energy is released. oxygen is used up. glucose is broken down. carbon dioxide is used up.
Answers: 1
You know the right answer?
How many atoms are in 25.00 g of B.
1.393 x 10^24 atoms of B
1.333 x 10^23 atoms of B
1.333 x 10^23 atoms of B
Questions


History, 16.11.2020 08:50



History, 16.11.2020 08:50

Mathematics, 16.11.2020 08:50





Physics, 16.11.2020 09:00

Chemistry, 16.11.2020 09:00

Arts, 16.11.2020 09:00

Mathematics, 16.11.2020 09:00

Chemistry, 16.11.2020 09:00


Mathematics, 16.11.2020 09:00


Mathematics, 16.11.2020 09:00

Physics, 16.11.2020 09:00