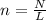Chemistry, 12.04.2021 02:20 mstrish71oteauw
How many moles are in 3.86 x 1023 molecules of Carbon Dioxide (CO2)?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
What is the correct term for living the most sustainable life you can within your current circumstances?
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

You know the right answer?
How many moles are in 3.86 x 1023 molecules of Carbon Dioxide (CO2)?...
Questions


Mathematics, 28.01.2020 16:04






Physics, 28.01.2020 16:04




Mathematics, 28.01.2020 16:04




English, 28.01.2020 16:04

Biology, 28.01.2020 16:04

Mathematics, 28.01.2020 16:04