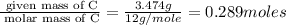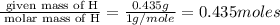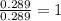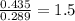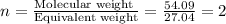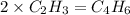Chemistry, 13.04.2021 02:00 staz13wiggins
When 3.915 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 12.74 grams of CO2 and 3.913 grams of H2O were produced. In a separate experiment, the molar mass of the compound was found to be 54.09 g/mol. Determine the empirical formula and the molecular formula of the hydrocarbon.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Initially, the balloon had 3.0 liters of gas at a pressure of 400 kpa and was at a temperature of 294 k. if the balloon is cooled to 277 k and its volume decreased to 1 l, what will the new pressure in the balloon be?
Answers: 1

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

You know the right answer?
When 3.915 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 12.74 grams...
Questions






Chemistry, 19.04.2021 02:20

Business, 19.04.2021 02:20


Mathematics, 19.04.2021 02:20

Mathematics, 19.04.2021 02:20



Mathematics, 19.04.2021 02:20


Mathematics, 19.04.2021 02:20

Mathematics, 19.04.2021 02:20




Mathematics, 19.04.2021 02:20

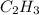 and
and 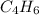 respectivley.
respectivley.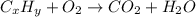
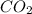 = 12.74 g
= 12.74 g
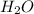 = 3.913 g
= 3.913 g
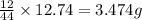 of carbon will be contained.
of carbon will be contained.
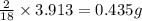 of hydrogen will be contained.
of hydrogen will be contained.