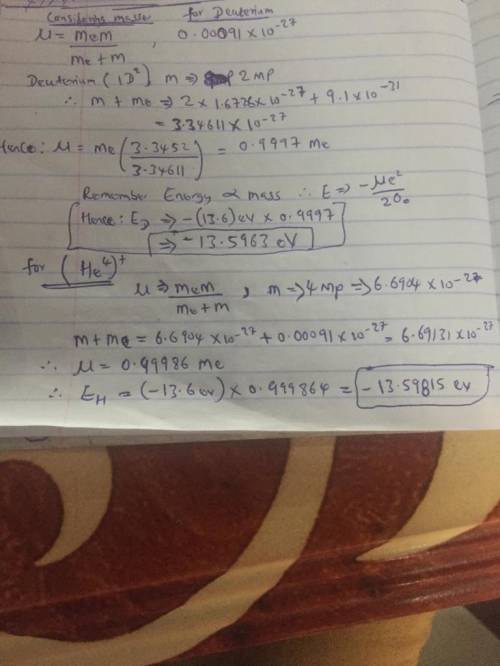Chemistry, 13.04.2021 03:00 kamnicole13
Hydrogen, deuterium, and singly ionized helium are all examples of one-electronatoms. The deuterium nucleus has the same charge as the hydrogen nucleus, and almost exactlytwice the mass. The helium nucleus has twice the charge of the hydrogen nucleus, and almostexactly four times the mass. Make a prediction of the ratios of the ground state energies of theseatoms, considering that current spectroscopy accuracy is extremely good (on the order of107).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2

Chemistry, 23.06.2019 02:30
What role does weathering have in shaping earth’s surface? a) it allows sediments to fall out of a medium. b) it sediments settle on a new surface. c) it breaks down older material into sediments. d) it transports sediments to a different location. will give brainliest, answer quickly.
Answers: 2

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1

You know the right answer?
Hydrogen, deuterium, and singly ionized helium are all examples of one-electronatoms. The deuterium...
Questions





Mathematics, 15.07.2020 23:01



Chemistry, 15.07.2020 23:01


Physics, 15.07.2020 23:01

Mathematics, 15.07.2020 23:01

Health, 15.07.2020 23:01

Mathematics, 15.07.2020 23:01


Mathematics, 15.07.2020 23:01





Mathematics, 15.07.2020 23:01