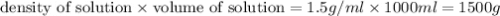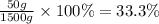Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:50
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2


Chemistry, 21.06.2019 22:00
If the particles in a sample of matter have an orderly arrangement and move only in place, the sample is a
Answers: 1

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2
You know the right answer?
What is the percent by mass concentration of 1000 ml of a solution (d=1.5 g/ml) that contains 50 g o...
Questions



Mathematics, 20.10.2020 16:01

Mathematics, 20.10.2020 16:01

Chemistry, 20.10.2020 16:01

Chemistry, 20.10.2020 16:01



English, 20.10.2020 16:01







Mathematics, 20.10.2020 16:01

Mathematics, 20.10.2020 16:01