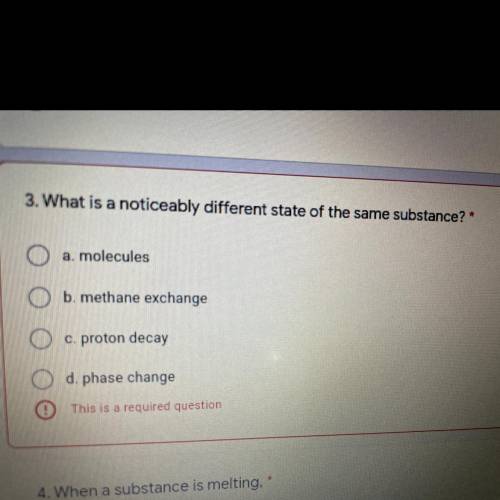
Help like rn please
...

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:50
Achemical reaction (also known as a chemical change) produces substances that are chemically different from the starting materials. an example of a chemical reaction is the formation of water from hydrogen and oxygen gas.in a physical change, a substance changes its physical appearance but not its chemical identity. an example of physical change is the formation of liquid water from solid water, a familiar process called melting. physically, liquid water looks very different from solid water (ice) but the chemical identity, water, is the same for both. which of following changes that affect the composition of our atmosphere involve physical changes and which involve chemical reactions? oxygen gas changes to ozone during thunderstorms carbon dioxide is produced by the combustion of gasoline in an automobile engine. when coal, oil, and natural gas are decomposed in landsills they produce methane gas. freezing rain develops when a warm air mass overrides a cold air mass. fog forms from water vapor when the temperature drops below the dew point
Answers: 1

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1
You know the right answer?
Questions

Mathematics, 28.11.2021 14:00





Business, 28.11.2021 14:00

Mathematics, 28.11.2021 14:00

World Languages, 28.11.2021 14:00

Computers and Technology, 28.11.2021 14:00


Physics, 28.11.2021 14:00


Biology, 28.11.2021 14:00

Business, 28.11.2021 14:00

Mathematics, 28.11.2021 14:00

Business, 28.11.2021 14:00

Physics, 28.11.2021 14:00

History, 28.11.2021 14:00

Mathematics, 28.11.2021 14:00

History, 28.11.2021 14:00